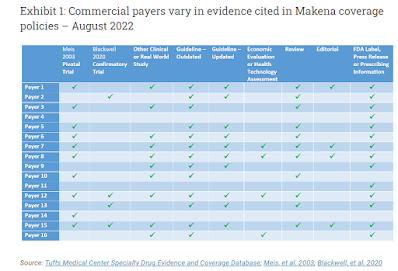
The Tufts health economics group has a new article out looking at how payers vary alot in the evidence they site for the same service. The service in question is coverage of "Makena," an accelerated-approval drug for pre-term birth risk reduction. Data in later studies has not held up well, and a FDA panel voted almost unanimously it should be withdrawn. (Any actual action is pending).
But here's why I'm flagging it. The Tufts group looked at how 9 data sources were cited, or not, by 16 different payers (a 9x16 data table). The point is, lots of payer variability. See the whole article open access at Health Affairs here.
Getting some buzz on Twitter is a September 2022 book, "Drugs and the FDA: Safety, Efficacy, and the Public's Trust," by Mikkael A. Sekeres (here). I understand the book provides a fly-by of historical FDA regulation, and then focuses on the battle over accelerated approval Avastin labeling around 2011.