This spring, I released a white paper (and an accompanying short video) on CMS policy and innovation (entry point here). One of my main points was that barriers to innovation can come from unexpected places and often, only those directly companies or stakeholders directly affected know about it.
Many lab tests come from the hospital outpatient environment, for example, from academically-based transplant clinics. Since about 2015, CMS has bundled lab tests associated with hospital outpatient clinic visits (or procedures) unless the test meets one of a few exceptions, the main one being it is a test of human DNA/RNA. Other categories lab tests are bundled, such as proteomic tests and slide-based tests and infectious disease tests (based on pathogen DNA/RNA).
There are several examples of this in the May 26, 2022 transmittal, "Update of Hospital Outpatient Prospective Payment System" (CR12761, online here.)
COVID Tests Exempt from Bundling
Pathogen (microbiology) tests are bundled, except, COVID tests are exempt from bundling. Tests that are exempt from bundling have "Status A" (paid on a separate clin lab fee schedule).
In the 26-page document, Table 1 lists COVID-related lab tests and shows they are assigned "Status A" separately payable, whereas other microbiology tests have bundled status. CMS also unbundles PLA codes that represent panel tests that include COVID (e.g. a 22-pathogen panel 0202U).
- Note that MACs are not required to cover large-panel pathogen tests and MolDx recently released an LCD that covers large pathogen panels only in exceptional circumstances. However, tests like 0202U are "unbundled" and payable in theory in the hospital outpatient setting.
0108U - Newly Test Freed from Bundling by ADLT Status
CMS has a special classification and payment category called ADLT, Advanced Diagnostic Laboratory Tests. There are two ways to get this. In both, you have to be a sole-source test. Method A is to be a MAAA-type test, and covered, and clinically unique. Method B is to be an FDA cleared or approved sole source test. (The law is meant to be an incentive to getting FDA clearance of your sole-source LDT test).
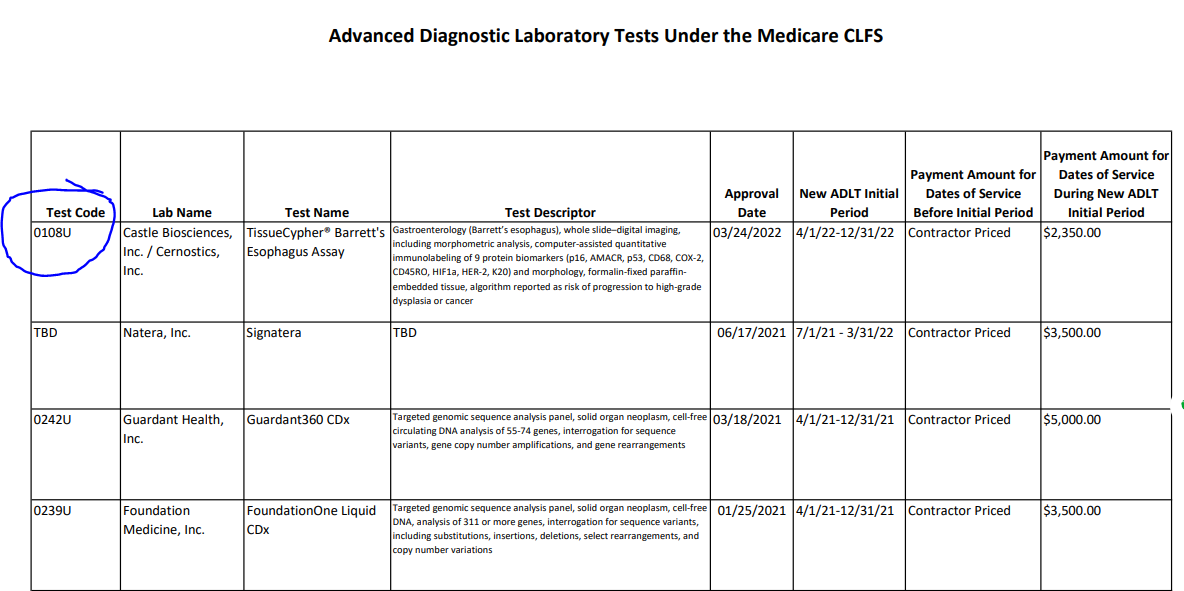
Test 108U was recently awarded ADLT status, and therefore, it's outpatient payment status is converted from bundled test to unbundled test (status A = unbundled). This is discussed in the transmittal.
In the ADLT sheet at CMS, here, CMS says that prior to setting an ADLT price of $2350 test 0108U was priced by contractor pricing. However, I think this is a bit wrong, since 0108U was on the CLFS fee schedule prior to ADLT status, not under contractor pricing. The former CLFS price was actually a bit higher at $2513. The ADLT price should represent "the lowest price available on the first day the test was marketed."
 |
| 2022 ADLT |
| 2021 CLFS |
Nerd Notes.
CMS has a webpage for the date of service rule, and a particularly interesting attachment there is a Zip file with an Excel of about 500 tests which meet an exemption from bundling (basically, human DNA RNA tests). This is like a filter of lab tests in OPPS Appendix B claims processing instructions for which the lab test status is "A" rather than "Q4".
From CLFS to CLFS-ADLT. I realize it is a little ridiculous to add a "nerd note" to something which is already so wordy, but anyway. I had thought that if an ADLT test was already set on a national CLFS price, it wouldn't reset to the initial list price. That is, I thought the initial list price rule for ADLT's was only used when there wasn't any "initial" fee schedule national price. However, 0108U did have a fully established CLFS price and nonetheless the ADLT assignment at "initial payment period" has occured, as shown in the ADLT screen shot.
Proteomic MAAA Tests. Type A ADLT tests must be multi biomarker tests of DNA, RNA, or protein. CMS must have categorized the glass immuno slides as "proteomic biomarkers" in order to meet this rule.
"Covered." Being an ADLT means the test is "covered." I'm not aware of a CMS LCD for 0108U (nothing at the CMS Coverage Database), but it must be covered under some circumstances. I believe there are now one or two PLA codes that incorporate "AI" into the test descriptor, while the 0108U is "computer assisted" but does not use the terms ML or AI.
Type A vs Type B ADLT and Bundling. Type A (MAAA) ADLTs are released from outpatient bundling, but FDA ADLT tests (Type B) are not released from bundling. However, most FDA ADLT tests have been DNA RNA tests anyway, which aren't bundled whether or not they have ADLT status.
COVID Code List. The list includes 87913, COVID, mutation analysis (variants), created in February 2022 by AMA. This Table 1 of the document and appears to list all COVID codes, from teh earliest (U0001) and (87635) to the latest (e.g. PLA codes for 20 targets one of which is COVID). All the COVID tests are assigned Status Indicator A meaning they are paid separately and not bundled (most other lab tests are bundled). However, two COVID codes appear to be missing, 87636 and 87637, which are COVID plus FLU A/B (amp probe) and COVID plus FLU A/B plus RSV. These are on the outpatient code table "Appendix B" as "A" codes. I wrote CMS to ask they they were missing.
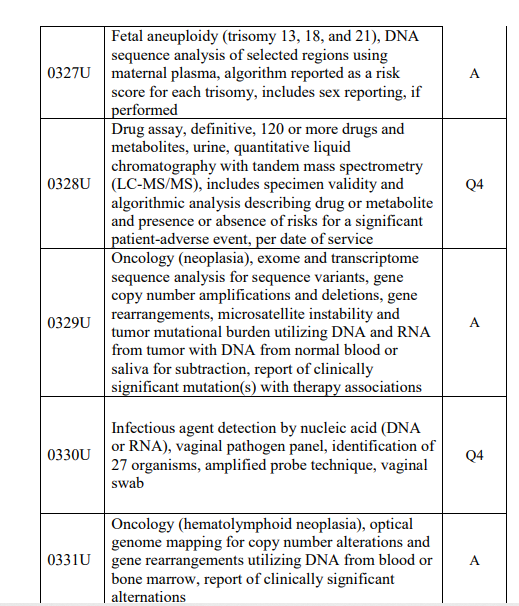
DNA-RNA Rule. Note in the table of new codes below, CMS unbundles (A) DNA RNA tests from human DNA RNA, but bundles other lab tests like 0328U drug assay and 0331U microbial.