One of the early landmark tests in precision medicine was the Oncotype DX test for breast cancer prognosis, first published in the NEJM in 2004. Today, there are a half-dozen tests recommended with a few variations in society guidelines.
ASCO has updated its guidance and published it April 19, 2022, as Andre et al., "Biomarkers for Adjuvant Endocrine and Chemotherapy in Early-Stage Breast Cancer" (30 pp). Tests include Oncotype, Mammaprint (FDA), Breast Cancer Index, Endopredict, Prosigna (FDA), IHC-4. When access to these is not available, they recommend Ki67 (a single immunostain). Find Andre' et al here, open access:
https://ascopubs.org/doi/full/10.1200/JCO.22.00069
The prior version was Harris et al. 2016.
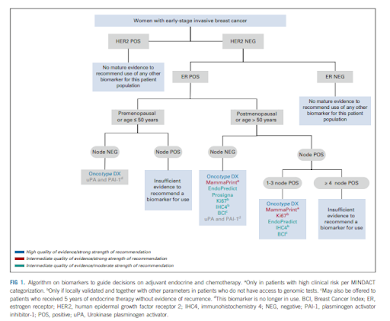 |
| Flow chart from Andre' 2022 |
See coverage at Genomeweb here.
History Lesson
Today, MolDx has several LCDs that are keyed directly to relevant extramural guidelines, such as tying PGx coverage to CPIC and FDA PGx recommendations.
Tidbit: In May 2016, MolDx briefly proposed, then withdrew, a plan to directly lock Medicare coverage directly to current ASCO recommendations. (My blog here). (One community objection, at the time, was that guidelines are only updated every 5-6 years, as we see here with Harris et al. versus Andre' et al, 2016, 2022).
The Brief Noisy History of "CDD" and CED
Not directly related, but my search for the above link led me first to a December 2016 blog where MolDx proposed withdrawing coverage for the Vectra test (here). That blog also talks about how MolDx seemed to be canceling or withdrawing references to its short-lived local "coverage with data development" or CDD effort. MolDx later dropped CDD paragraphs from the MolDx handbook in 2018, here.
A 2022 article on Medicare and risk sharing arrangements including CMS CED by Chen & Carlson tallied up a large number of those long-defunct MolDx local CDD policies (here). Not touching on MolDx CDD, but taking a deep dive into CMS NCD CED, see Zeitler et al. (entry point here).
Next: AI for Breast Cancer Prognostics
By keyword search, artificial intelligence and machine learning are not mentioned in Andre' et al. However, there are already not just original studies but quite extensive review articles on using machine learning for breast cancer prognostics (see review by Li et al, PLOS ONE, 2021, here; see also Yousif 2022 here, Wang 2021 here, Fitzgerald 2021 here.)
A Sociologist's View
See a book on precision medicine from the sociology and anthropology viewpoint, with a chapter on the adoption and changing viewpoints of the Oncotype Dx test. Chapter 2, Genomic techniques in standard care: Gene-expression profiling in early-stage breast cancer, in book: Personalized Cancer Medicine, Kerr et al., Manchester Univ Press, 2021. (Hardcover only).